| Lure of riches fuels testing
'To wash people out from their medication, to take away
at kind of treatment, that to me is inhumane.'
By Robert Whitaker, Globe Correspondent,
11/17/98
Third of four parts
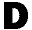 uring the first three weeks she spent in Fairview Riverside
Hospital in Minneapolis, Susan Endersbe, a 41-year-old woman struggling
with schizophrenia and suicidal impulses, obtained care and medication
that made her feel much better. uring the first three weeks she spent in Fairview Riverside
Hospital in Minneapolis, Susan Endersbe, a 41-year-old woman struggling
with schizophrenia and suicidal impulses, obtained care and medication
that made her feel much better.
Her mood lightened. By May 26, 1994, she was telling nurses that she
felt she'd be ready to leave soon.
But the very next day, she was referred to psychiatrist Faruk
Abuzzahab, an encounter that put her on a path that led to her death.
Abuzzahab, a past president of the Minnesota Psychiatric Society and
onetime chairman of its ethics committee, had a contract with Abbott
Laboratories to test the experimental antipsychotic drug Sertindole. What
he earned has not been disclosed, but such contracts typically pay
physicians much more than regular health insurance reimbursement, creating
a powerful incentive to put patients into commercial research trials.
Endersbe's death offers a peek into the financial side of
corporate-funded research of new drugs for the mentally ill. While the
development of new antipsychotic drugs has brought new hope for
schizophrenic patients and allowed many to live successfully in the
community, the clinical testing of these medications is also big business,
with the pursuit of money often in conflict with good patient care. The
result, in schizophrenia research, is a landscape tarnished by the greed
of some rogue investigators and repeated instances of patients being
harmed.
''The abuses are there,'' said Dr. Morris Goldman, associate professor
of psychiatry at the University of Chicago School of Medicine, who
investigated Abuzzahab's transgressions. ''A big part is the dollars
involved. ''
There are ''people drawn into this field because they are interested in
dollars,'' Goldman added. ''They are very profit-conscious. And that
combination of a lot of money, plus the added ethical dilemma you face in
human research, that is a bad combination. And there are particular risks
with psychiatric patients, with the whole issue of informed consent. It
can really go wrong.''
When Abuzzahab enrolled Endersbe into his drug trial, he later admitted
to state investigators, he ignored study criteria that excluded patients
who were suicidal. And as soon as she stopped taking the venlaxafine that
had controlled her symptoms, she quickly worsened, state investigators
reported.
Endersbe repeatedly told nurses that she intended to kill herself.
Devils were now struggling for her mind, her brother said. Her complaints
were recorded in nurses' notes, but Abuzzahab presented a rosy picture in
his research records for Abbott. Endersbe was experiencing no side effects
from the drug, he wrote, so he kept her in the trial.
For nearly two weeks, until June 8, Endersbe received no antipsychotic
medication. Then, on June 11, when she had been on the experimental drug
for three days, Abuzzahab granted her a day pass to leave the hospital
unaccompanied. She went to her apartment to gather some keepsakes, slipped
the key back under the door, and walked straight to the Franklin Avenue
Bridge. Just as she had said she would, Endersbe clambered over the
railing and leapt to her death in the Mississippi River.
''For nearly 20 years, my sister was managing to win the battle for her
survival, and when she went on a drug study, there were supposed to be
safeguards in place to protect her,'' said her brother, Edward Endersbe.
''Not only were they not in place, they neglected to have the usual
safeguards that she would have had if she stayed on as an inpatient''
outside the drug trial.
''And to wash people out from their medication, to take away any kind
of treatment, that to me is inhumane,'' he added. ''If they did that to
someone with a physical illness, I would think it would be criminal.''
Troubling questions about patient safety
People with schizophrenia, studies have shown, are at greatest risk of
suicide during a relapse, at the beginning of an acute psychotic episode,
and when they are discharged from the hospital. Taking part in a drug
trial often exposes them to all those factors.
In the 1990s, three antipsychotic drugs for schizophrenia have been
brought to the market: Zyprexa (olanzapine), Risperdal (risperidone), and
Seroquel (quetiapine). Abbott Laboratories also submitted an application
for Serlect (sertindole) - the drug Susan Endersbe was given - but
withdrew it after a Food and Drug Administration advisory committee
questioned its safety.
The results of the clinical trials submitted to the FDA, obtained by
the Globe through Freedom of Information requests, raise troubling
questions about the safety of schizophrenic patients enrolled in
commercial drug trials:
Among 12,176 patients from the US and abroad who had participated in
trials for all four drugs at the time the data was submitted to the FDA,
there were 88 deaths, including 38 suicides. [The figure includes deaths
up to 30 days after patients left the trial.] That is an overall death
rate of 1 of every 138 volunteers.
The suicide rate for the clinical trials is two to five times higher
than the norm. In the medical literature, annual suicide rates for
schizophrenia patients range from two to five deaths per 1,000 people. The
annual suicide rate in the trials, on a time-adjusted basis, was close to
10 per 1,000.
In trials for the three approved drugs - Zyprexa, Risperdal, and
Seroquel - 60 percent of the 7,269 patients who received the experimental
drug dropped out before the end of the study period, typically six to
eight weeks. Patients dropped out because the drugs didn't work, because
of side effects, and sometimes because they refused to continue.
No one has made a detailed study of the mortality or suicide rates of
subjects in schizophrenia drug trials. When queried, a half dozen
psychiatric researchers expressed varying responses to the numbers
collected by the Globe. Some thought they were remarkably high. Some
argued that the majority of the people entering the trials had been
chronically ill with schizophrenia, and thus a number of suicides and
deaths could be expected. And some researchers begged off; they did not
know what to make of the statistics.
Reasearchers seduced by lure of lucrative rewards
The research enterprise that has produced new neuroleptic drugs for
schizophrenia has paid off, of course, in important ways. The standard
drugs used from the 1950s to the early 1990s typically made patients feel
like zombies and caused major side effects that disrupted muscle control.
The antipsychotic drugs that have reached the market in recent years
[Risperdal, Zyprexa and Seroquel] are less sedating and appear to have
fewer harmful side effects.
Desperately ill schizophrenics have turned to clinical trials for early
access to these new drugs, and many of those patients attest to the
attentive care that they received from researchers conducting the trials.
But there are powerful financial factors at work that can tempt
researchers to enroll mentally ill patients into drug trials even when it
will not serve the patients' interests.
Antipsychotic drugs are considered a major and fast-growing market for
the pharmaceutical industry. In 1997, US sales topped $1.5 billion, double
the figure from two years earlier. Pharmaceutical companies developing
antipsychotic medications know that every day shaved from the testing
process can get them to the market a day sooner, and each day earlier to
market represents $1 million to $2 million in additional sales per drug.
The companies need to get patients into their trials quickly, and they
will pay researchers handsomely for doing so. But they also expect the
researchers to deliver. Researchers who do not meet their patient quotas
are not likely to get a contract for the next project.
''The pressures are enormous,'' said Newell Unfried, executive vice
president of the Alliance for Multispecialty Research, a group of leading
for-profit research centers. Increasingly, he added, performance reviews
''are being drawn up by pharmaceutical companies on every physician in
clinical research on their patient recruitment. There is pressure to get
these patients.''
In fact, the drug-testing industry today is a highly competitive,
profit-driven enterprise. Only about 25 percent of drug trials are
conducted in nonprofit academic medical centers. The rest are conducted by
community physicians who contract with drug makers or by for-profit
companies whose sole focus is conducting drug trials.
Physicians who do commercial drug research full-time regularly report
generating more than $1 million in annual revenues, and profits exceeding
$300,000.
In the 1990s, a Providence-based company, Clinical Studies, built a
nationwide chain of research centers focusing on drugs for Alzheimer's
disease, schizophrenia, and other neurological disorders. The founders
sold it in 1997 to a physician-practice management company for stock
valued at $85 million. Hoping to make similar scores, venture capital
groups have poured more than $100 million into start-ups seeking to
duplicate Clinical Studies' success.
In this environment, researchers know that the ''product'' they deliver
is patients who can be enrolled in trials and kept there until they
complete the study protocol. Physicians often earn bonuses for meeting
patient-recruitment goals.
Critics say the monetary incentives put researchers on a slippery
slope, particularly when it comes to testing schizophrenia drugs. Their
financial interests push them to recruit patients aggressively, yet the
trials expose the patients to numerous risks.
But Dr. Stanley Cheren, a Brookline psychiatrist who conducts
commercial drug trials on a full-time basis, insists ''there are a number
of things that keep you sober and honest - your own conscience.
''Leaving that aside, you are accountable to the patient and to the
patient's family, and they do raise hell when things aren't going well.
The hospital staff keeps you accountable. And the third thing always
lurking in the background is the FDA. If you recruit a lot of patients and
do it fast, the FDA takes that as a red flag and audits your site.''
However, the financial pressures and opportunities have been known to
lead physicians astray. A criminal case in Georgia revealed just how far.
Dr. Richard Borison and Bruce Diamond were longtime favorites of
pharmaceutical companies developing schizophrenia drugs. Borison, 48,
chairman of the psychiatry department at the Medical College of Georgia in
Augusta, and Diamond, 53, a pharmacologist on the school's faculty, had
demonstrated time and again a knack for bringing psychotic patients
quickly into trials funded by drug companies. Eli Lilly, Janssen, Zeneca,
Novartis - they all came knocking.
As faculty, Borison and Diamond were supposed to get approval from the
medical school for their research. Drug-company payments for clinical
trials were supposed to be sent to the school. But according to Georgia
state authorities, who indicted the pair in early 1997, they began in 1989
to have the companies send payments directly to them. They opened an
office across the street from the medical school, used a commercial
service to do ethical reviews of their studies, and placed their staff on
the school's payroll while keeping all the research funding for
themselves.
From 1989 to 1996, Borison and Diamond made over $10 million from drug
trials, including more than $4 million from schizophrenia drugs, according
to the indictment and sworn testimony from their staff during an
investigation by the Augusta Veterans Affairs Hospital, where Borison was
chief of psychiatry. Witnesses said the two researchers focused on getting
patients into the studies and paid minimal attention to their care.
To recruit the mostly male patients, they hired attractive young women,
who later testified that they were paid bonuses that ran into the
thousands of dollars. According to testimony, one staffer was given a
Honda Accord for coaxing schizophrenia patients into trials.
To earn these rewards, workers phoned and later met mentally ill
patients who were stable and living in the community and offered them $150
to check themselves into the VA hospital so they could be in a study.
Patients already in locked wards were offered cigarettes for agreeing to
participate.
''When there is a possibility that you are going to get a car, you're
going to do whatever you can,'' study coordinator Angela Touhey told VA
investigators.
Touhey and the other study coordinators, many of whom had no medical
training, determined whether a patient belonged in a study. According to
an FDA investigation, untrained staff drew blood samples and adjusted
doses of the experimental drugs. Touhey said Borison and Diamond hardly
saw patients during the trials. At one weekly meeting, Touhey said Diamond
told the staff they weren't interested in hearing about the patients.
''Bruce told me, 'We don't care about how the patients are doing. We
just want to know how many people you have enrolled in the past week or
couple of weeks,''' Touhey said.
The two researchers lived high, according to Georgia authorities. They
tucked away more than $5 million in cash and securities, drove
Mercedes-Benz cars, and dropped more than $400,000 on antiques.
Borison bought medieval tournament armor from Sotheby's auction house
in New York, and mahogany-paneled doors in London, for an
11,000-square-foot castle that he was planning to build. The indictment
charged that they bribed a staff member, Terri Davis, to keep quiet after
a schizophrenia patient in an olanzapine trial attempted suicide.
''This whole thing was very dirty,'' Dr. David Hess, chief of neurology
at the VA, told the hospital's investigators. ''It was basically a numbers
game. These patients are purely used for the greed of the researchers.
That was very apparent to me what was going on.''
Diamond and Borison are now behind bars. In December, 1997, Diamond
pleaded guilty to theft and bribery charges and was sentenced to five
years in prison, fined $125,000, and ordered to pay $1.1 million to the
college.
After one week of testimony at his trial in October, Borison pleaded
guilty to theft and a racketeering charge. He was sentenced to 15 years in
prison, fined $125,000, and ordered to pay $4.26 million to the college.
Researcher caught falsifying records
When the Minnesota Board of Medical Practice received allegations that
Dr. Faruk Abuzzahab was endangering the welfare of patients in his general
psychiatric practice, it hired Dr. Morris Goldman of the University of
Chicago to investigate. One of the first things he discovered was that
Abuzzahab was fudging records in order to put patients into lucrative drug
studies.
''He would have the patient's diagnosis called one thing in the regular
chart, and then the person would be put on a drug study and the person's
diagnosis would be called something else to fit the criteria,'' Goldman
said in an interview.
In July, after Abuzzahab admitted to the board that he had entered
''disturbed and vulnerable'' patients into drug studies even though they
didn't meet eligibility criteria, and had ''kept them in the study after
their conditions deteriorated,'' the medical board suspended his license.
Among other things, Abuzzahab admitted that he cycled one woman with
paranoid schizophrenia through three drug trials from 1991 to 1994,
despite the fact that she did poorly on the experimental drugs and
complained of being a guinea pig. Eventually she became catatonic and
incontinent; she was then given standard therapies and responded well.
In another instance, the psychiatrist took a woman off clozapine, a
newly approved drug that had led her to make a remarkable recovery, and
enrolled her in a drug trial. The woman had previously spent 13 years as
an inpatient at a psychiatric institution, but while on clozapine she had
been able to go into the community and even hold a job. One day she
approached Abuzzahab with questions about clozapine's side effects; he
immediately stopped the drug that had helped her so much and put her into
an olanzapine trial. She deteriorated until she found her way to another
physician, who put her back on clozapine.
According to the state medical board, Susan Endersbe was another
patient whom Abuzzahab ''recklessly'' entered into a clinical trial. The
doctor, the medical board noted, entered her into the trial the first day
he met her.
''I think a key flaw in this whole thing is the financial gain,'' said
Edward Endersbe. ''I am not a communist, but it is really disturbing to
see how much this is driven by financial gain, and that it can, and does,
such harm. There is no question in my mind that that is what happened with
my sister. My sister very much wanted to live and to be a survivor.''
Incentives encourage aggressive recruiting
Abuzzahab and Borison can be dismissed as isolated ''bad apples'' who
unfairly tarnish the image of all researchers who conduct schizophrenia
drug trials. But the economic incentives they pursued so eagerly are in
play in all commercial drug testing.
Dr. Angela Bowen, president of the Western Institutional Review Board,
a commercial ethics review service, said researchers are becoming more
aggressive in their use of advertising to recruit patients into
schizophrenia studies. Research centers that don't have any ongoing
studies have even submitted advertisements for IRB approval seeking
depressed and schizophrenic patients who would consent to being withdrawn
from their medications, she said. The researchers apparently wanted to
develop a pool of mentally ill people, already off standard therapies, who
could be quickly enrolled once new drug contracts are secured.
''We, of course, don't approve those ads,'' Bowen said. ''It boggles
the mind.''
Aggressive recruitment of schizophrenic patients is problematic because
participating in drug trials exposes them to the risk of relapse and
suffering. To determine whether a drug can curb psychosis, it must be
tested in people who are actively psychotic. That scientific standard,
which is endorsed by the FDA, leads straight into an ethical minefield.
One approach is for researchers to seek people who are already actively
sick, either because they have stopped taking medication on their own or
because the symptoms aren't being adequately controlled by medication. But
many of these patients suffer from ''disorganized thoughts'' and some
degree of psychosis, making it questionable whether they can give
meaningful informed consent.
Another route is to seek stable patients who are on medication. These
patients, however, must be hospitalized and their medications stopped in
order for them to take part, with the expectation that their delusions and
hallucinations will return. The patients must become sick again to be
useful subjects.
''If you don't take people who have reestablished active disease, then
you don't know what you are looking at'' when you test the drug, said
Robert Temple, associate director for medical policy at the FDA's Center
for Drug Evaluation and Research. ''That is why you have to have a
washout. And once you realize that you have to do medication withdrawal,
you have already gone into what people are worried about, and that is
giving a relapse.''
But Temple said the profession has not ''reached the conclusion that
allowing people to withdraw [from medication] is dangerous to them, if
they are closely monitored and well informed.''
If patients become too ill when their regular drug is stopped,
researchers may shorten the washout period. They then enter the active
part of the study and either receive the experimental drug, a standard
drug [usually Haldol], or an inactive placebo. Researchers will also use
''rescue medications'' if the patients deteriorate too rapidly.
''People do not have to become dramatically, floridly more psychotic in
order to be ready to go into the study,'' said Nina Schooler, director of
psychiatry research at Hillside Hospital, a division of Long Island Jewish
Health System in Queens. ''We are not talking about requiring dramatic
psychotic relapse.''
Still, the essence of the process is to let the patients become sicker.
''The patient is suffering,'' acknowledged Cheren, the Brookline
psychiatrist who conducts drug studies. ''Psychosis makes a person suffer.
A second [risk] is that the patient could become more violent and
destructive to himself and to others. Third, people think that acute
psychosis does damage to the brain, and you want to limit those periods.
Multiple episodes of acute psychosis lead to poorer long-term outcomes.
You want to limit all of those things as much as possible.''
Stopping a schizophrenic patient's medication raises two other
concerns. In normal clinical care, when a patient is taken off drugs, the
recommended practice is to lower the dosage gradually, because abrupt
withdrawal has been shown in numerous studies to dramatically increase the
risk of relapse. Moreover, evidence is emerging that sudden withdrawal may
lead to a rebound psychosis more severe than if the patient had never been
treated.
''It is not simply a matter of not being treated and going back to
baseline of an untreated state,'' said Dr. Ross Baldessarini, professor of
psychiatry and neuroscience at Harvard Medical School. ''There is an
additional risk involved that may be due to the discontinuation itself
.... We are trying to get people sensitized to this issue.''
David Cohen, a professor of social work at the University of Montreal
who has done extensive research on neuroleptic drugs, said abrupt
withdrawal can also lead to flu-like symptoms, nausea, and vomiting. This,
he said, is a withdrawal reaction, not just a return of previous symptoms.
''There will be new symptoms the person has never experienced before,''
Cohen added. ''But they don't tell them the kind of suffering it will
entail. They don't tell the patients how severe these symptoms may be, and
that they could be life threatening, and that they could be driven to kill
themselves.''
There are, of course, potential benefits for the research subjects. The
primary one is that they may respond to the experimental drug, and be able
to continue to get it by participating in a long-term followup study. But
the mentally ill patients can expect, at least at first, to go through a
difficult period.
''I know that when I was three or four days into the washout I was
really struggling,'' said D.L., a 41-year-old woman in Illinois diagnosed
with mild psychotic disorder who recently entered a trial of an
experimental antipsychotic. ''My symptoms increased dramatically. It was
getting scary. If they had been any more severe, I know that I couldn't
have handled it. I hope that if that had happened, I would not have hurt
myself. But when you are delusional, it is hard to know what you are going
to do.''
Researchers grapple with ways to reduce patient risk
Within the psychiatric research community, there is controversy over
whether drug studies can be designed to reduce the risk to patients. In
Europe, for instance, researchers are reluctant to conduct
placebo-controlled trials for schizophrenia. Instead, European researchers
usually conduct trials that compare the experimental drug to a standard
therapy.
That is also how US drug trials are typically designed for medical
conditions such as high blood pressure, for which there are already
effective approved drugs. But in schizophrenia research, the FDA believes
that patients' reactions to antipsychotic drugs are so variable that
without a placebo group, the tests cannot show whether an experimental
drug is effective. That could result in ineffective drugs being approved,
said the FDA's Temple.
''You have to think about the people in the trial, but you also have to
think of the consequences to the community of marketing ineffective
antipsychotics,'' he said.
Others find that justification unsatisfying.
''Can you imagine an antibiotic study that would compare the
experimental drug to a sugar pill?'' asked Unfried, head of the alliance
of for-profit research centers. ''Can you imagine that goofy study? With
the result that the patient died from too much infection? In those
studies, you use a standard therapy as a basis for comparison. The FDA has
a role to protect people and to make sure you design the trial in such a
way that you do not hurt people.''
Testing violations lead to a death in California
Anyone agreeing to take part in a risky drug trial would expect
attentive medical care. But that wasn't the case with Borison and Diamond.
It wasn't the case with Abuzzahab. And it was not what Jennifer Abigayle
McIntyre received when she entered a trial in 1993 for Janssen
Pharmaceutica's experimental drug risperidone.
After she died, an investigation by the California Department of Health
found violations that took 16 pages to detail.
In the spring of 1993, Abigayle's mother, psychiatrist Judith Vukov,
accompanied Dr. Robert Liberman, director of schizophrenia research at the
University of California, Los Angeles, to lobby Congress to fund research
on mental illness. Her daughter had schizoaffective disorder [a variant of
schizophrenia], existing medications weren't helping her, and, as Liberman
had told her, experimental treatments offered the best hope.
Later that same summer, on July 20, 1993, according to the California
Department of Health and court records, Abigayle McIntyre was transferred
to the research ward at Camarillo State Hospital, where doctors were
treating patients with experimental protocols developed by UCLA
researchers.
Abigayle signed an informed-consent form that stated she would be given
a physical exam, be closely monitored, and that if her condition worsened,
her treatment would be ''promptly adjusted.'' In fact, the investigation
showed, she never received a physical, nor did the staff prepare any plan
for treating her psychiatric and behavioral problems.
The principal researcher, Dr. Barringer Marshall Jr., gave her a new
diagnosis, chronic schizophrenia, which made her eligible for the
risperidone study.
As her previous medications were withdrawn, she became more volatile,
crying and screaming for hours on the phone with her mother. On Aug. 12,
as part of the protocol, she was prescribed Haldol, a standard
antipsychotic. Even though she began to suffer terrible headaches and her
blood pressure soared, she was kept on Haldol. The protocol called for her
to take the drug for three weeks before she could be switched to
risperidone.
Her mother complained, but to no avail.
''Just because I am a doctor doesn't mean anything,'' Vukov said. ''If
you have a psychiatric child, your opinion is useless.''
The last time the unit's staff made an entry on Abigayle's chart was
Aug. 26. On Sept. 11, her mother took her out of the hospital on a day
pass. At some point her daughter swallowed a large number of aspirin; she
died several hours later after emergency room physicians failed to
promptly diagnose the aspirin poisoning.
''I thought research was the best treatment in the country,'' said
Vukov. ''Today it is not. It is the most dangerous. Abigayle wouldn't have
gotten sick like that if she hadn't been in research. She ended up outside
my control and outside the control of a good doctor, who would have done
something about [her illness]. She was actively neglected in research.''
Messy picture often buried in a marketing glow
When a drug company submits data to the FDA, it must detail all deaths
that occurred in the clinical trials, along with a full accounting of side
effects, patients who dropped out of the study, and any other problems
suffered by the patients.
But that often messy picture is rarely reflected in the accounts of the
drug's performance that get published in medical journals.
Zeneca's Seroquel, approved by the FDA last fall, is the newest
antipsychotic drug on the market. When the Globe compared the mixed
results hidden in FDA files, including 14 deaths, with the glowing
conclusions published in medical journals, the differences were stark.
According to the FDA's review of Zeneca's data, 80 percent of the 2,162
patients given Seroquel in clinical tests stopped taking the drug before
the trial was over, even though the trials often lasted less than eight
weeks. Of those who dropped out, 1,033 did so because the drug didn't help
them, 398 simply refused to continue or were lost to follow-up, and 86 had
to stop because of adverse reactions that included excess sleepinees,
convulsions, suicide attempts, and depression.
But when articles began appearing in medical journals on the results in
the summer of 1997, the authors glossed over the dropouts, failed to
report on the deaths, and drew rosy conclusions. An article in the
Archives of General Psychiatry hailed Seroquel as ''an effective
antipsychotic with a favorable safety profile''; a report in Biological
Psychiatry touted the drug as ''well tolerated and clinically effective in
the treatment of schizophrenia. ''
''Most of the trial results that are published have as authors both
[researchers] and people involved with the trial who work for the
company,'' noted New York-based researcher Schooler, who Zeneca designated
to be its spokesperson. ''Of course people put the most positive spin
possible on their results.''
One result of this marketing spin is that practicing psychiatrists
often say they no longer know what to believe about new drugs. Although
Seroquel and the other new drugs do appear to cause fewer side effects
than standard antipsychotics, there are still many questions about how
they compare in efficacy.
''What gets put out is not necessarily the truth,'' Dr. E. Fuller
Torrey, director of research for the National Alliance for the Mentally
Ill, said this summer at the group's annual conference. ''You'd be
surprised at how much money many of my colleagues are regularly taking
from the pharmaceutical industry. $10,000 is not an uncommon amount to be
given for giving a talk.... If we really hope to answer the question of
new antipsychotics vis-a-vis the old ones, we're going to need something
other than the pharmaceutical industry to provide funds on it.''
Tomorrow: Vulnerable and unprotected
This story ran on page A01 of the Boston Globe on
11/17/98.
© Copyright 1998
Globe Newspaper Company.
| ![]()
![]()
![]()
![]()

